Scope Statement
To define the necessary requirements (including metadata) that will drive the identification and harmonization of standards to facilitate the collection of supplemental EHR-derived data.
This initiative will develop and validate a standards-based data architecture so that a structured set of data can be accessed from EHRs and be stored for merger with comparable data for other relevant purposes to include:
The electronic Case Report Form (eCRF) used for clinical research including Patient Centered Outcomes Research (PCOR)
The Incident Report used for patient safety reporting leveraging AHRQ ‘Common Formats’ and FDA form 3500/3500a
The Surveillance Case Report Form used for public health reporting of infectious diseases
The collection of patient information used for Determination of Coverage, as resources permit.
The infrastructure will consist of four new standards that will enable EHRs to capture and store structured data. These will consist of:
- a standard for the CDEs that will be used to fill the specified forms or templates;
- a standard for the structure or design of the form or template (container);
- a standard for how EHRs interact with the form or template; and
- a standard to enable these forms or templates to auto-populate with data extracted from the existing EHR
The standards will facilitate the collection of data in such a way that any researcher, clinical trial sponsor and/or reporting entity can access and interpret the data in electronic format. They will also support development of concise, architectural guidance using easy-to-understand documentation, user-friendly tooling and formal models to assist vendors in applying technical requirements for the customized use of specified forms or templates. For the purposes of this initiative, the data collected will not be stored within the EHR system. In sharing this data, ONC recognizes that certain forms of data may be subject to particular state or federal laws regulating use and disclosure. Standard specifications will incorporate the tools necessary for driving interoperability such as XML and the CDISC/ IHE integration profile Retrieve Form for Data Capture (RFD) ; this does not, however, imply any constraints on data formats that can be used during data capture and processing, as long as they do not prevent interoperability. The RFD integration profile is currently used within the research community to embed structured electronic forms with common data elements within the EHR to facilitate collection of research data. The SDC Initiative will align with and leverage other initiatives of ONC . It will also build upon external initiatives that are focused on improving the comparability and utility of data derived from independent collection efforts through standardizing definitions of data elements and tools, such as PROMIS, PhenX, caDSR, and other initiatives identified in the NLM-NIH Common Data Element repository, and the AHRQ ‘Common Formats’ and Electronic Data Methods (EDM) Forum for Comparative Effectiveness Research (CER).
Value Statement
Given the significant Federal investments made in EHR adoption in the last 4 years, structured data capture within EHRs is poised to be a critical component of a variety of health services, quality measurement and clinical and health services research. Stage 3 Meaningful Use (MU) will focus on creating a learning health system to support quality, research, and improve public and population health. This initiative will lead the national vision to design the trusted mechanisms to enable patient information to flow securely from the system it was collected—the EHR—to other systems, such as research consortia, registries, bio repositories and public health systems, with an authorized use for it. Information will be shared in compliance with policy, regulation, and Patient Consent Directives (e.g., 42 C.F.R Part 2 Confidentiality of alcohol and drug abuse patient records; and 38 USC § 7332-Confidentiality of certain medical records). The identification and harmonization of standards for structured data capture within EHRs will not only help achieve this vision, but they will also help reduce the:
Data collection burden on health care providers by enabling secure, single-point data entry that populates to multiple systems
Need to make site-specific modifications to EHR system capabilities in order to enable participation in important reporting and research activities
Barriers to volunteer adverse event reporting on medical products to public health agencies leading to improvements in population health
These efforts will identify a standard for structured data, whether it is used for a clinical trial, Determination of Coverage, or to report on a patient safety event, which can be collected in a timely manner, then readily compared and aggregated improving the overall quality, value and utility of these data. Furthermore, the development of a national infrastructure will improve access to standardized electronic versions of data collection instruments relevant for use in research and patient care such as validated instruments for collecting data on pain, fatigue, physical function, depression, anxiety and social function. It will be easier to integrate these instruments into EHRs in ways that will ultimately reduce duplicate data entry. Likewise, data collected will be more comparable and therefore more useful in ascertaining what works best for different patient populations.
Target Outcomes & Expected Deliverables
The SDC Initiative will provide an infrastructure to standardize the capture and expanded use of patient-level data collected within an EHR. In the short term, specification of standards for data reuse will support and spur development and implementation of software and pilots that will inform refinement of these standards, prior to their consideration for inclusion in Meaningful Use and EHR certification requirements. In the longer term, the additional functionality will support enhancements and efficiencies in such diverse domains as patient-centered outcomes research and clinical trials, adverse event reporting and public health monitoring and surveillance, Determination of Coverage and patient care.
The value of this initiative will be measured through the attainment of the following immediate and long-term outcomes:
- Identification of functional requirements from a Use Case describing key conditions and business rules to enable the capture and storage of specified forms or templates, while protecting privacy and confidentiality
- Development of concise consensus-driven architectural guidance using easy-to-understand documentation, user-friendly tooling and formal models to assist researchers, patient safety personnel, software vendors and others in applying technical requirements for the customized use of specified forms or templates. Guidance will be updated and versioned appropriately to allow ubiquitous access to all parties.
- Execution of one or more pilots to evaluate the use of the specified form standard in specific contexts, such as patient-centered outcomes research and patient safety event reporting. The pilots will examine the application of specified-form standard for the conduct of PCOR and patient safety event reporting, drawing upon the NLM/NIH common data elements repository, as well as AHRQ ‘Common Formats’ for patient safety events.
- Proliferation and use of NIH-identified and curated CDEs for PCOR and AHRQ ‘Common Formats’ for patient safety event reporting
- Development or identification of four national standards specific for: CDEs used to fill electronic forms or templates, the structure or design of the form or template, the standardized functions for how EHRs interact with those standards, and the specifications that enable these forms or templates to auto-populate with data extracted from the existing EHR.
- Alignment and integration to other health IT infrastructure (through a Learning Health System) to support effective maintenance, distribution, and use of specified forms or templates
- Enhancement of patient care through improvements in quality and safety interventions, population health, and research
- Improvement in provider experience and workflow when using EHRs for patient care and other purposes
Standards
Standardization efforts established by other projects will be leveraged. These include, but are not limited to:
Stakeholders and Interested Parties
Stakeholders and interested Parties include but are not limited to the following:
- Healthcare Providers and Clinical Informaticians
- Clinical/PCOR Research Community and CER/PCOR Thought Leaders and organizations, such as: Patient-Centered Outcomes Research Institute (PCORI), EDM Forum, CDISC, Electronic Medical Records and Genomics Network (eMERGE), Distributed Ambulatory Research in Therapeutics Network (DARTNet), Electronic Patient-Reported Outcome (ePRO) Consortium, ASTER-D
- Patient Safety Organizations (PSOs)
- Privacy and Security Experts
- Patient Advocates
- Pharmaceutical Firms
- Device manufacturers
- Government Agencies:
- Food & Drug Administration (FDA), Assistant Secretary for Planning and Evaluation (ASPE), NIH (NLM & other Institutes/Centers), AHRQ, Centers for Disease Control (CDC), Centers for Medicare & Medicaid Services (CMS), Indian Health Services, Human Resources and Services Administration (HRSA), Institute of Medicine (IOM), Veterans Administration (VA), Department of Defense (DoD), Social Security Administration (SSA), Department of Transportation (DoT), State Medicaid Programs
- Vendors:
- EHR/EMR systems, Health Information Exchange (HIE), Data Warehouse/Data Mart, Electronic Data Capture (EDC) and Patient Safety Event Reporting System
- Standards-Related Organizations: Standards Development Organizations (SDOs), vocabulary/terminology organizations, standards setting organizations
- Healthcare payers, particularly those with robust research, quality improvement (QI), patient safety and public health activities, including professionals involved in registries and surveillance at a reginonal and national level
- Professional liability carriers
- Healthcare Professional associations
Proposed Timeline
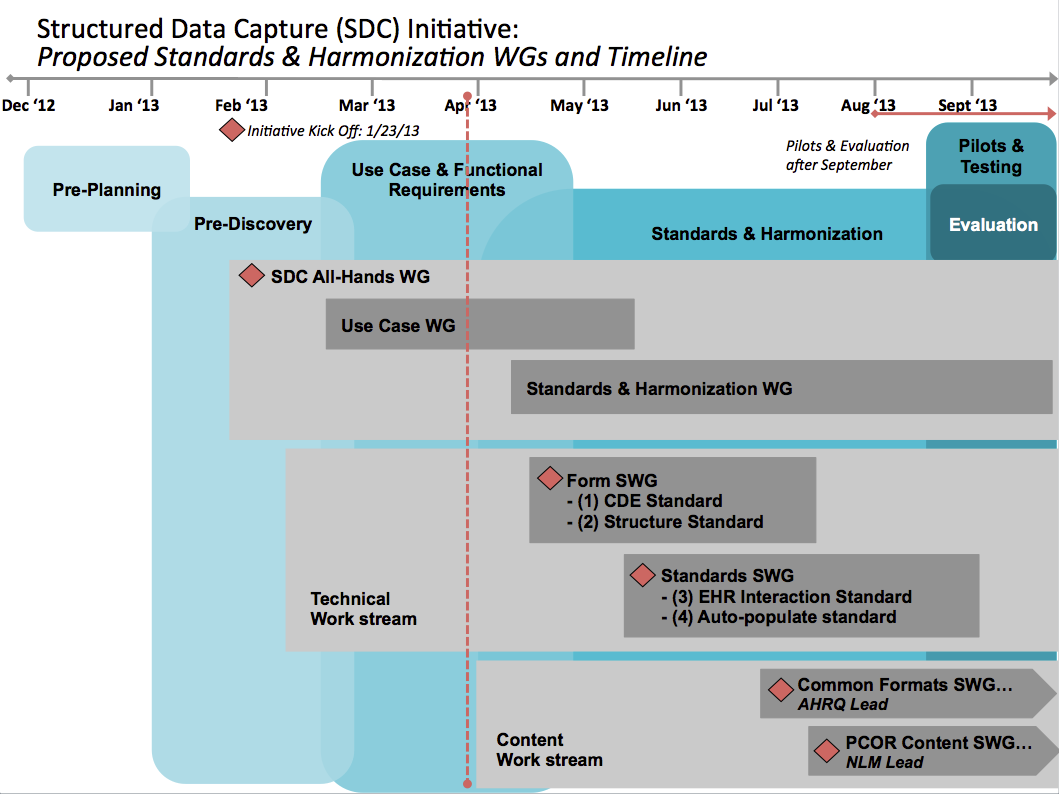
This timeline represents notional estimates of planned activities and will be adjusted as work moves forward through the initiative phases.
Risks
- Identification of content models for standard that do not compete or overlap with existing or developing models
- Insufficient engagement and participation by vendor communities, and eventual adoption in Certified EHR products
- Insufficient commitment by various vendors and organizations to participate in SDC pilots
- Accessibility of a CDE library or repository in time for pilots and completion of pilots in time for future stages of Meaningful Use
- Competing definitions of Common Data Element and CDE libraries
- Identification of a useful but parsimonious set of data elements for forms/templates that can auto-populate with data extracted from the existing EHR in time for pilots
- Overwriting and/or duplicate data entry
- Solutions for research, patient safety, and public health reporting may vary; one standard solution may not apply
- Standards and solutions may not scale to small vendors and small practices
- Proposed project timeline does not reflect actual deadlines in relevant standards or regulatory bodies
- Not all EHR systems offer or can support pre-population functionality; if the auto-populate standard is prescriptive, the SDC timeline may be delayed as vendors implement the functionality in their systems
- Providers may not ‘fill’ all CDEs displayed on an electronic form
- Accuracy of EHR data and reliability of diagnosis codes for reuse in clinical research
- Patient privacy: de-identification of EHR data for clinical research
Success Metrics and/or Success Criteria
- Upfront engagement in development of use case requirements and solution from potential pilot sites
- Balloted standard(s) are included as EHR certification criteria
- Proliferation and use of NLM-identified and curated CDEs for PCOR and AHRQ ‘Common Formats’ for patient safety event reporting
- Harmonization of CDEs among federal agencies and research communities in other countries
