Initiative Overview and Scope
For detailed information on initiative goals, objectives and intended outcomes, please see the SDC Project Charter.
Summary: The Structured Data Capture (SDC) initiative will develop and validate a standards-based data architecture so that a structured set of data can be accessed from EHRs and be stored for merger with comparable data for other relevant purposes to include:
- The electronic Case Report Form (eCRF) used for clinical research including Patient Centered Outcomes Research (PCOR)
- The Incident Report used for patient safety reporting leveraging AHRQ ‘Common Formats’
- The Surveillance Case Report Form used for public health reporting of infectious diseases
- The collection of patient information used for determination of coverage, as resources permit.
Following is a visual conceptualization of the initiative workflow.
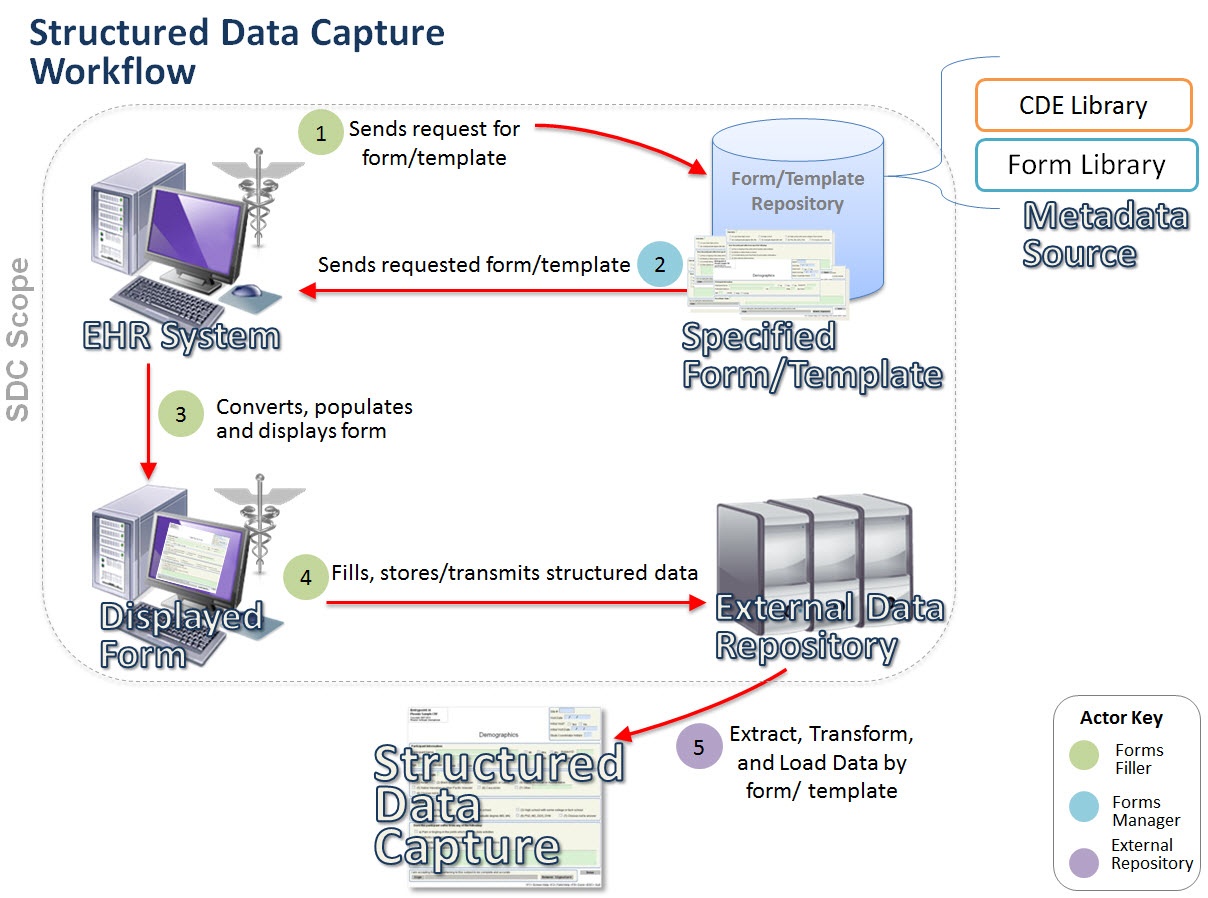
The proposed infrastructure will consist of four new standards that will enable EHRs to capture and store structured data. These will consist of: a standard for the CDEs that will be used to fill the specified forms or templates; a standard for the structure or design of the form or template (container); a standard for how EHRs interact with the form or template; and a standard to enable these forms or templates to auto-populate with data extracted from the existing EHR. The standards will facilitate the collection of data in such a way that any researcher, clinical trial sponsor and/or reporting entity can access and interpret the data in electronic format. For more information about this initiative, please see the
Structured Data Capture Charter.
